By Bradley Deline
Editor’s Note: Brad is an Associate Professor in the Geosciences Department of the University of West Georgia. He earned his BS in Geology at the University of Michigan and MS and PhD at the University of Cincinnati.

In 1989, Stephen Jay Gould published his book, Wonderful Life, describing the fossils of the Burgess Shale as well as their importance in understanding the history of life on Earth. He proposed that these weird wonders (such as the animal shown in Figure 1) from the ancient sea represented a zenith in morphologic disparity. Disparity is a measure of the diversity of forms or features found within a group of organisms. In other words, how different are animals from each other. He proposed that life experimented with many different body plans, some of which worked and diversified through time and others failed and went extinct. Therefore, the animals of today are constrained within the body plans of the Cambrian survivors.
For almost thirty years this hypothesis remained largely untested. This was the result of how difficult it is to mathematically compare very different organisms. For example, how similar is a fish to a housefly or a sponge or a seastar? If we could map the evolution of bodies across the animal kingdom this would allow us to answer some fundamental questions about life over the last half billion years.
Back in 2009, I was discussing these ideas at a conference over drinks with Philip Donoghue (University of Bristol) and Kevin Peterson (Dartmouth College) and we formulated a plan to tackle this long-standing hypothesis. Phil proposed we use the recently published work of German anatomist Peter Ax (1996, 2000, 2003). Ax systematically described different animal groups from bilaterians to butterflies listing out the features that defined those groups. From this resource we were able to quantify the anatomy of almost 400 animals based on over 1800 characteristics. We then mapped how all of those characters were related so we could code characters as non-applicable, for example coding the limb structure doesn’t make sense for a sponge and we wanted to capture that in the analysis. We then included 80 fossil animals (along with over a hundred new characters from those animals) that would include the range of body forms found during the Cambrian Explosion. The vast majority of these fossils are exquisitely preserved, but half of the characters are still features that have never been seen in the fossil record (for example cellular structures). To correct this, we built an evolutionary tree that incorporated all of the modern and fossil animals. We then used this tree to statistically model evolution to estimate the unpreserved characteristics of the ancient animals.
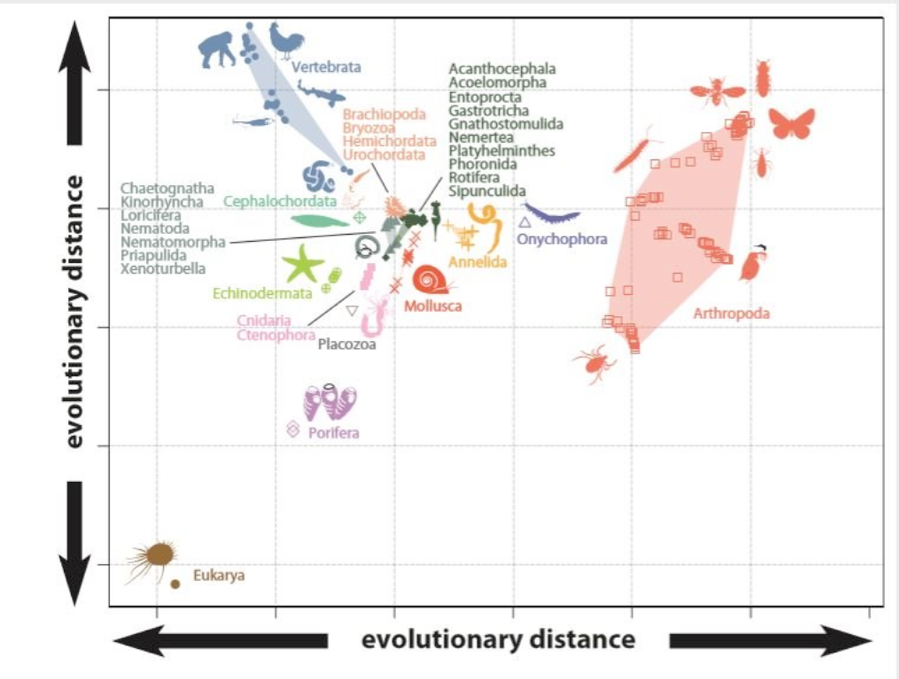
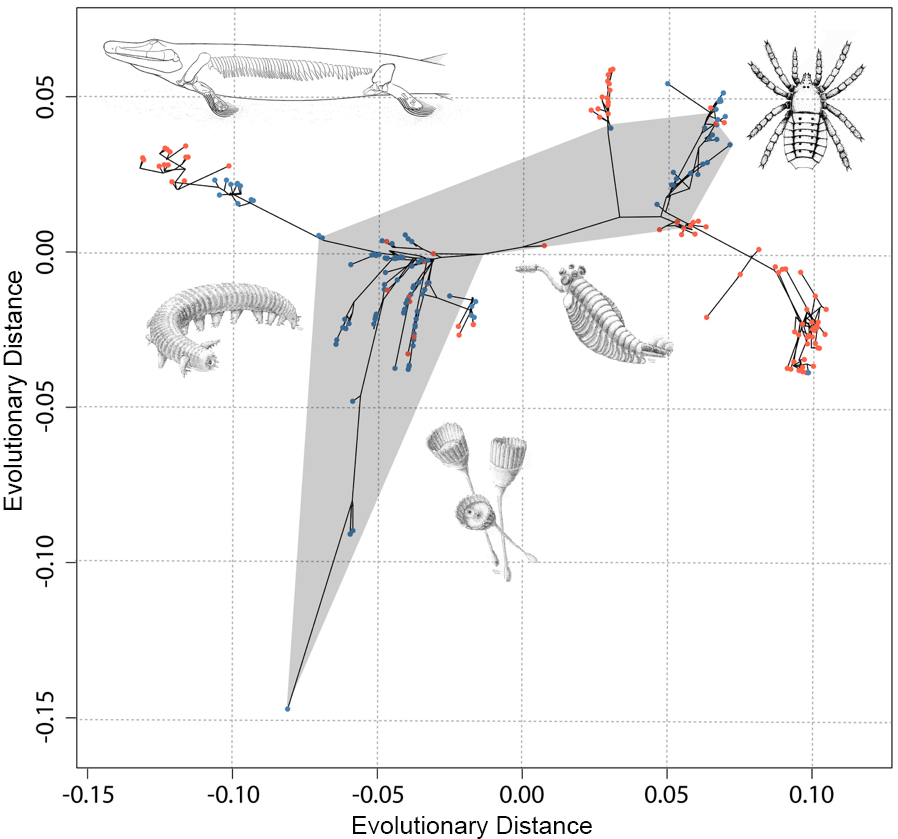
The resulting evolutionary map of animal body plans can be seen in Figure 2. Animals that plot closely together share features giving them an overall similar appearance. Many phyla cluster closely together in the central area of the shape space based on their shared wormy appearance. The extremes of the evolutionary space are occupied by arthropods, such as insects, spiders, and crabs, and vertebrates, such as fish, reptiles, and us. When we included the Cambrian oddities (Figure 3) they plotted in the intermediate areas between modern groups. The implication of this is that modern animal groups appear distinctive because of the extinction of transitional forms rather than rapid early evolution. Also, this means that the weird animals from the Cambrian only appear bizarre because they are unfamiliar compared with what we have in the world today. In other words, today’s world is filled with many more weird wonders compared with the ancient ocean. The evolution of body plans wasn’t restricted to the Cambrian, but life kept exploring new forms through time. These expansions often correlated with major evolutionary events, such as the transition onto land as seen in vertebrates, mollusks, annelids, and arthropods (Figure 3).
We also wanted to explore the underlying mechanisms for this expansion through time. To do this we collected data on genome lengths as well as the diversity of proteins and regulatory genes for living animals. We found that the distribution of animals in anatomical space correlated with genome length and regulatory genes, but not the type or diversity of proteins. This indicates that the controls on the timing and expression of genes were key to animal evolution.
Overall, this study produces a new prospective on animal biodiversification and the mechanisms that have led to the diversity of life today.
References
Ax P (1996) Multicellular Animals: A New Approach to the Phylogenetic Order in Nature(Springer, Berlin), Vol 1, p 225.
Ax P (2000) Multicellular Animals: The Phylogenetic System of the Metazoa (Springer,Berlin), Vol 2, p 369.
Ax P (2003) Multicellular Animals: Order in Nature–System Made by Man (Springer,Berlin), Vol 3, p 317.
Dunlop JA (1996). Systematics of the fossil arachnids.Revue suisse de Zoologie, hors série, 173-184.
Gould SJ (1989) Wonderful Life: The Burgess Shale and the Nature of History (Norton, New York).
Shubin NH, Daeschler, EB, & Jenkins, FA (2014). Pelvic girdle and fin of Tiktaalik roseae. Proceedings of the National Academy of Sciences, 111(3), 893-899.